The Regulatory Maze: Why Smart Tattoos Are a 'Wild West' for Regulators

Ever felt technology moves so fast, laws can barely keep up? That feeling is spot-on for mood-adaptive smart tattoos. This cutting-edge tech operates in a largely undefined regulatory space. Such a landscape creates palpable excitement. It also fosters significant uncertainty. Users and manufacturers both feel this.
Smart tattoos blur established lines. Are they medical devices? Perhaps wellness gadgets. Or simply advanced fashion statements? This core ambiguity challenges traditional regulatory frameworks. AwareInk.tech's analysis highlights a crucial point: legal definitions often trail far behind technological leaps. Regulation is inherently reactive. This reactive nature creates a vacuum. New technologies like smart tattoos occupy this void. It's a genuine 'Wild West' scenario, creating both opportunity and risk.
So, what are the implications for you, the user? Without clear rules, trusting safety claims becomes difficult. Data privacy questions loom large. This regulatory lag introduces distinct uncertainties. Understanding any existing oversight, however limited, is therefore crucial. It empowers informed decision-making. We will delve into these specific complexities.
You might also find this interesting: content disclaimers
FDA's Lens: Classifying Smart Tattoos in the USA (Medical Device vs. Wellness)
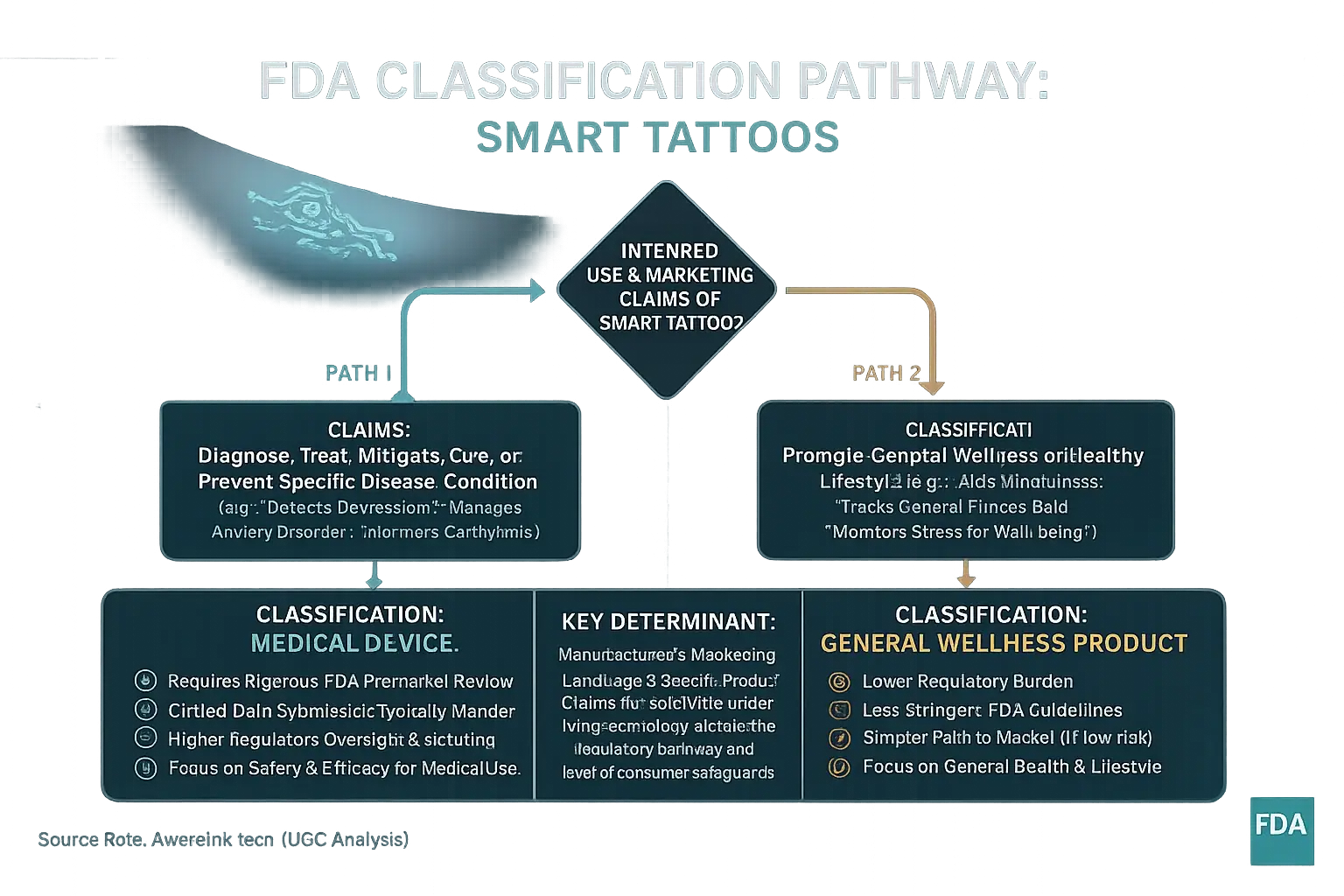
The U.S. Food and Drug Administration (FDA) regulates health-related products. FDA classification depends heavily on a product's intended use and its marketing claims. Products fall into two main categories: medical devices or general wellness products. This distinction is critical for mood-adaptive smart tattoos. It directly impacts the level of regulatory oversight.
Smart tattoos' classification hinges on their specific advertised functions. A tattoo claiming to diagnose, treat, or prevent a disease becomes a medical device. Examples include assertions of detecting depression or managing anxiety disorders. Such devices require rigorous FDA premarket review, including clinical data. Conversely, a tattoo marketed to aid general mindfulness likely qualifies as a general wellness product. These products encounter less stringent FDA guidelines. User safety remains a consideration. But the path to market is simpler.
A medical device classification generally offers users more protection. This protection stems from FDA-validated performance and safety claims. General wellness products receive less direct FDA scrutiny before reaching consumers. AwareInk.tech analysis reveals an important, often unspoken truth here. A manufacturer's marketing language, not just the core technology, frequently dictates the regulatory pathway. This choice significantly influences the level of consumer safeguards. Understanding these claims is key for you.
Beyond the USA: CE Marking and European Compliance for Smart Tattoos

CE marking indicates product conformity for the European Economic Area (EEA). This is mandatory . The mark itself declares compliance with essential health, safety, and environmental protection standards. Understand this: it is not a quality award. It is the manufacturer's official statement. Consumer trust across the EEA builds upon this declaration.
So, how does CE marking impact smart tattoos? Europe differentiates. Smart tattoos with clear medical purposes fall under the stringent Medical Device Regulation (MDR) 2017/745 . This path requires extensive conformity assessments, often involving Notified Bodies . Purely wellness-focused smart tattoos might navigate other directives. For instance, the Radio Equipment Directive (RED) 2014/53/EU governs wireless communication capabilities , while RoHS restricts hazardous substances.
Manufacturers primarily shoulder CE marking responsibility. Here's the often unspoken part: many products achieve CE marking through manufacturer self-declaration. They compile and maintain the necessary technical documentation, including risk assessments and validation data . This system places significant trust in their diligence. AwareInk.tech's analysis highlights this crucial distinction from some pre-market approval systems, impacting how users might verify product claims and safety assurances.
Your Emotional Data, Your Rules: Privacy Laws (GDPR & CCPA) for Smart Tattoos
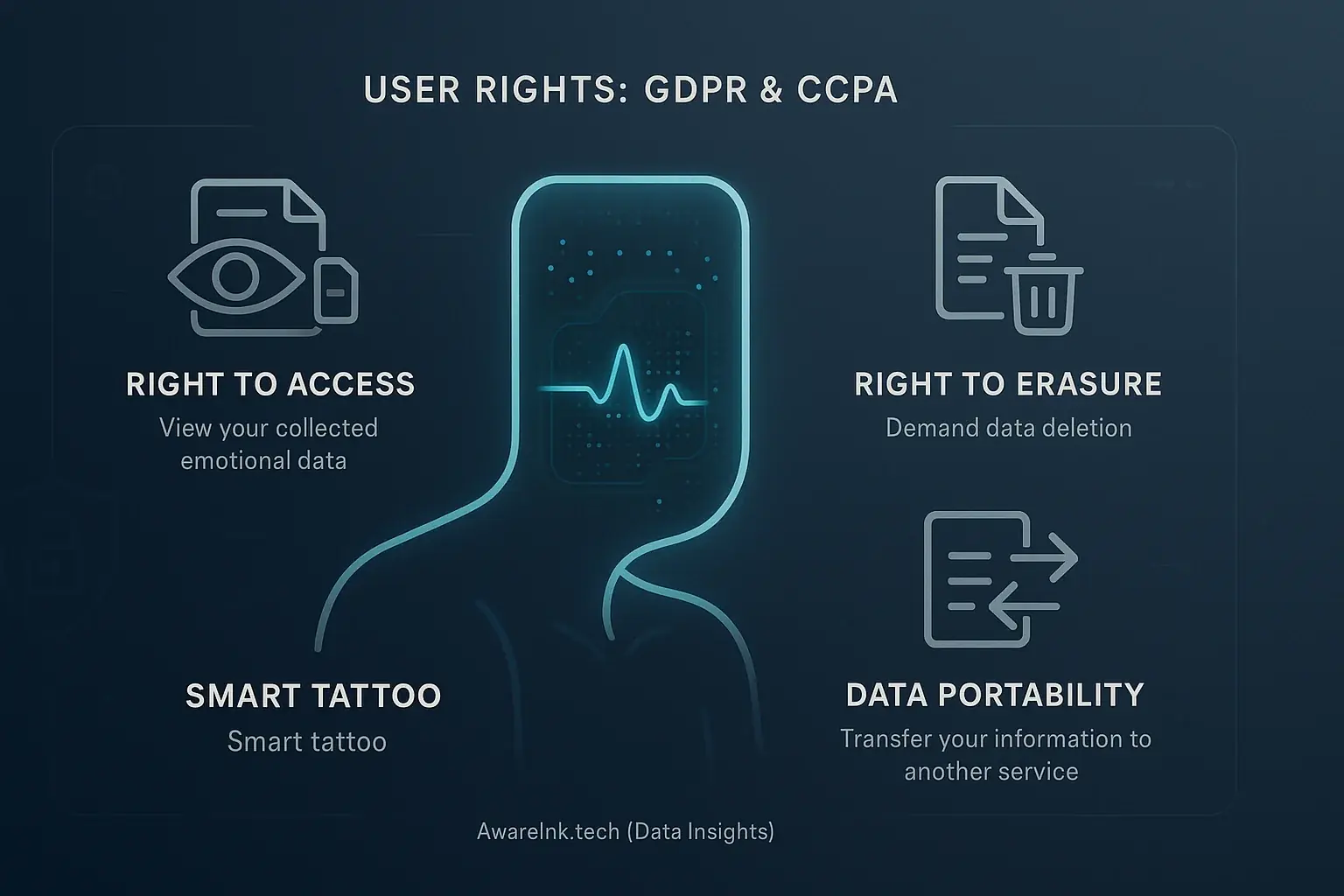
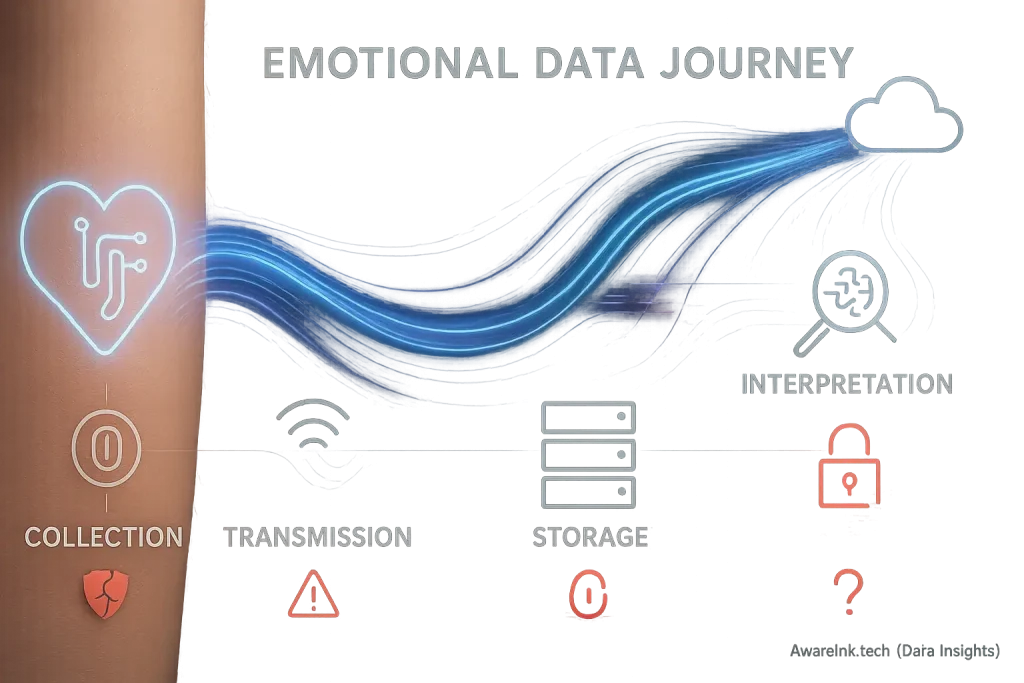
Mood-adaptive smart tattoos collect highly sensitive personal data. This data reveals your emotional states. Data privacy laws like GDPR in Europe and CCPA in California directly address this. These regulations exist to protect your information. They grant you significant power over your personal data. This protection is a vital safeguard.
You possess several key rights under these important laws. The 'Right to Access' allows you to see your collected emotional data. You can demand its deletion through the 'Right to Erasure'. Data Portability even lets you transfer your information to another service. Manufacturers must honor these requests promptly. These rights clearly empower you, the user.
Companies face strict obligations regarding your emotional data. They must obtain your explicit consent before any data collection. Robust security measures are mandatory to protect this intimate information. Transparency about how your data is used is also a firm requirement. Here is an unspoken truth: these laws achieve their full protective power when you actively understand and assert your rights. Knowing these rights helps you shield your unique emotional blueprint. AwareInk.tech will guide you in asking the crucial questions to ensure your privacy.
Skin Deep: Biocompatibility Standards for Smart Tattoo Materials
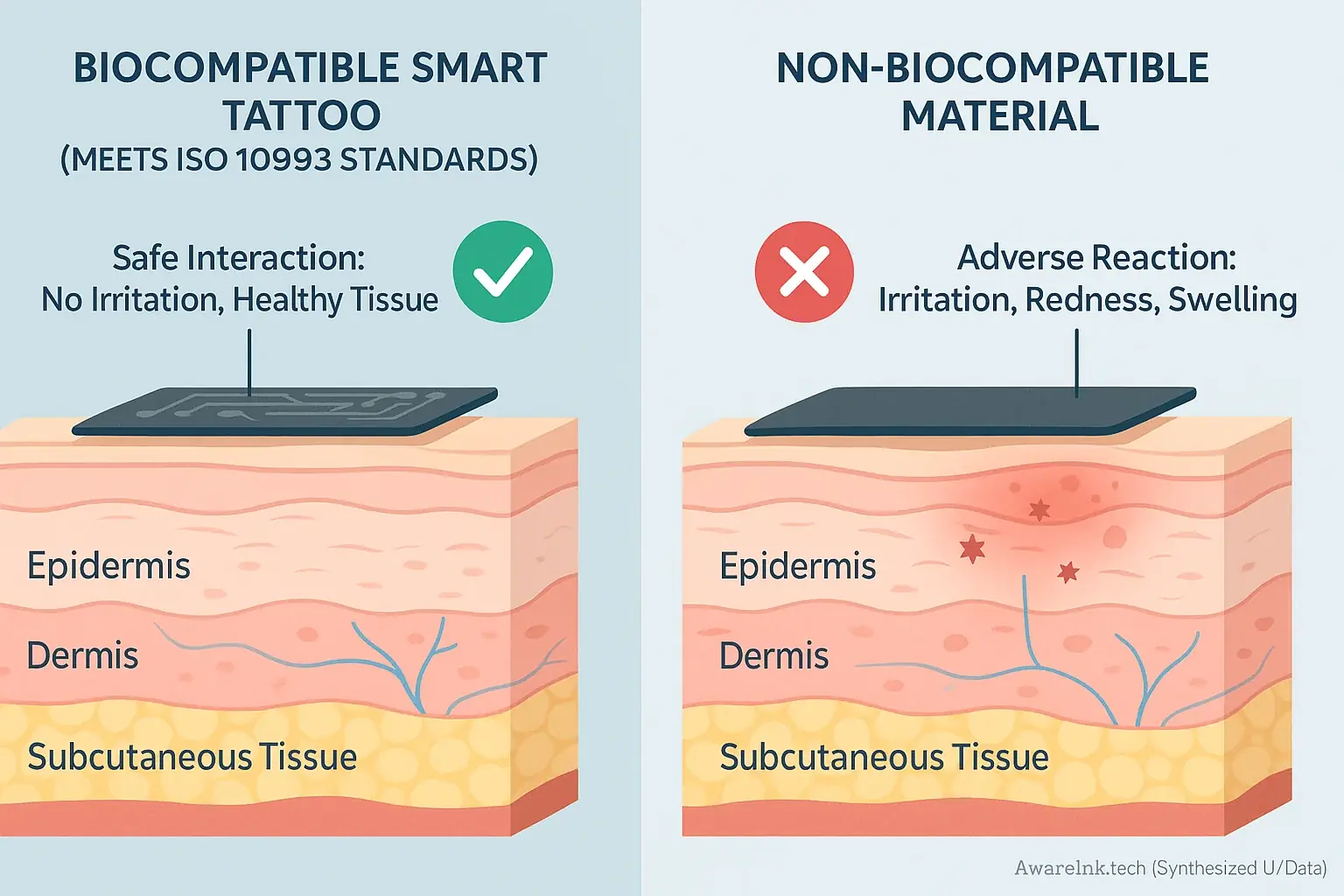
Biocompatibility defines material safety. This safety is critical for direct skin contact. Smart tattoo materials absolutely require this assurance. The materials must not cause adverse reactions. User safety depends on it.
Biocompatibility standards, such as ISO 10993, mandate rigorous material testing. This testing identifies potential toxicity using methods like in vitro cell exposure. It also assesses skin irritation risks through direct application tests. Sensitization tests check for allergic responses, often using animal models. All smart tattoo components, like conductive inks, flexible substrates, and adhesives, undergo these evaluations. Manufacturers bear responsibility for proving material safety against these established benchmarks.
A crucial insight emerges from AwareInk.tech's research. Even 'biocompatible' materials can sometimes cause issues with extended use. Long-term wear introduces new factors. Individual skin sensitivities also greatly influence outcomes. This means vigilance is key.
What does this mean for you? Users must monitor their skin closely. Look for persistent redness. Note any unusual swelling. Address any itchiness that develops under or around the tattoo. This proactive vigilance is essential for a positive long-term experience.
Looking Ahead: Future Regulatory Trends & Challenges for Mood Tech

Smart tattoo technology rapidly advances. Future regulation must evolve alongside it. New complexities will certainly surface. Advanced our system for interpreting emotions presents one such area. Global data sharing protocols create another. These domains urgently require new, thoughtful regulatory frameworks. AwareInk.tech sees these trends clearly.
Regulators will face substantial future challenges. Addressing user indicates bias in emotional analysis demands careful attention. Data interoperability across international borders presents another hurdle. Standardizing biocompatibility for novel nanomaterials remains a complex task. The unspoken truth? Regulation often follows problems. Proactive policymaking proves difficult. AwareInk's analysis confirms this pattern.
Collaboration becomes essential for navigating this future. Manufacturers, users, and policymakers must engage actively. This collective effort fosters responsible technological innovation. It helps craft regulations protecting users without stifling progress. AwareInk.tech commits to tracking these key regulatory developments. We will share emerging insights with you.